NuvaRing and Deep Vein Thrombosis Lawsuit
Attorney Brenda Fulmer of the Mass Tort Unit at Searcy Denney filed a products liability lawsuit today against the manufacturers of NuvaRing.
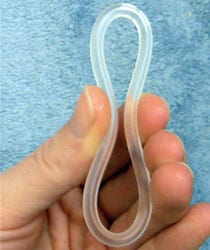
NuvaRing is a novel medical device that is used for contraceptive. It has become a popular product used by young girls and women due to the convenience of not having to take a birth control pill each day. The NuvaRing product, which is a plastic ring embedded with ethinyl estradiol and the progestin etonogestrel, is inserted for 3 weeks and then removed for 7 days each month.
This latest lawsuit was filed on behalf of a 27 year old Palm Beach County woman who suffered a deep vein thrombosis and pulmonary emboli after using the product for several years.
 Currently, there are approximately 800 individual lawsuits filed across the country by girls and women who suffered blood clot-related injuries after use of NuvaRing. All of the NuvaRing cases filed in the federal court system are coordinated for pre-trial purposes through multi-district (MDL) proceedings in St. Louis, Missouri. The allegations of these lawsuits contend that Organon and Merck (as the successor to Schering Plough Corporation), the manufacturers and distributors of NuvaRing, have failed to provide proper warnings and safety information to patients and their gynecologists regarding the unique risks faced by women who use this implantable contraceptive device, which utilizes a third-generation progestin. There is strong evidence that third generation progestins, such as desogestrel, gestodene, and etonogestrel, are far more dangerous than the older, second generation progestins such as levonorgestrel, which have been on the market for far longer.
Currently, there are approximately 800 individual lawsuits filed across the country by girls and women who suffered blood clot-related injuries after use of NuvaRing. All of the NuvaRing cases filed in the federal court system are coordinated for pre-trial purposes through multi-district (MDL) proceedings in St. Louis, Missouri. The allegations of these lawsuits contend that Organon and Merck (as the successor to Schering Plough Corporation), the manufacturers and distributors of NuvaRing, have failed to provide proper warnings and safety information to patients and their gynecologists regarding the unique risks faced by women who use this implantable contraceptive device, which utilizes a third-generation progestin. There is strong evidence that third generation progestins, such as desogestrel, gestodene, and etonogestrel, are far more dangerous than the older, second generation progestins such as levonorgestrel, which have been on the market for far longer.
Similar allegations are being more in more than 10,000 individual lawsuits over Yaz, Yasmin, Ocella, Beyaz, and other newer birth control pills which utilize the fourth generation progestin drospirenone, as are, too, considered far more dangerous than second generation progestins. Neither third generation nor fourth generation progestins provider greater protection against unwanted pregnancy than the safer second generation progestins, but the newer progestins have garnered greater market share due to a minimization of the risks and the fact that the newer progestins are less likely to cause the androgenic or testosterone-effects associated with second generation progestins.
The lawsuits alleging that NuvaRing is defective also point to a concern that the device might not deliver consistent levels of hormones, a concern that also extends to other birth control products that use novel delivery methods, such as the Ortho-Evra birth control patch.
Share This


