Third Ethicon Bellwether Trial Set for March 2015
Judge Goodwin is presiding over seven different federal multidistrict litigation established for similar cases filed against various medical device manufacturers, including American Medical Systems, Boston Scientific, Coloplast Corp., Cook Medical, Ethicon , C.R. Bard, and Neomedic.
Each of the complaints involves allegations by women who experienced painful complications following the use of mesh for transvaginal repair of pelvic organ prolapse and/or stress urinary incontinence.
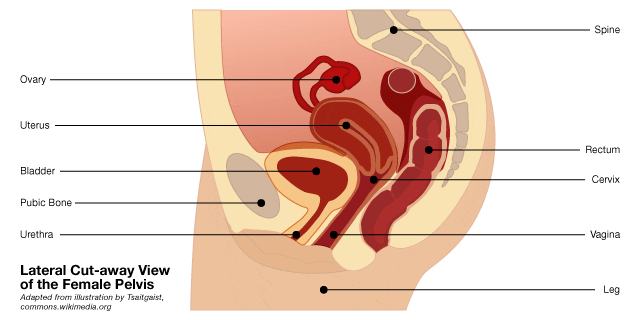
Cut away of the female pelvis. When the muscles (in the pelvic floor) supporting the bladder are stretched or torn, the resulting condition is known as “pelvic organ prolapse” or POP, which generally requires surgical treatment sometimes with the use of a medical device known as a transvaginal mesh.
As part of the coordinated pretrial proceedings, Judge Goodwin has ordered that groups of cases against several different manufacturers be prepared for trial dates, known as “bellwether” cases because the outcomes are designed to help the parties determine how juries may respond to certain evidence and testimony that may be repeated throughout the litigation.
Earlier this year, the first Ethicon bellwether trial, involving a lawsuit filed by Carolyn Lewis, ended in a defense verdict after Judge Goodwin entered judgment as a matter of law following presentation of the plaintiff’s case. Judge Goodwin determined that Ms. Lewis failed to present sufficient evidence that the Ethicon TVT mesh was defectively designed or caused her to suffer an injury. In September 2014, a second bellwether trial, involving an Ethicon bladder sling product ended in a $3.25 million jury verdict for the Plaintiff.
The third bellwether trial set to begin early December has been postponed until March 2015. The Bellew trial was the last in an initial series of three bellwether cases involving women who experienced complications with Ethicon mesh.
The allegations raised by Ms. Bellew are typical of those brought in other claims, showing that she suffered a long list of injuries due to the defective design of the Ethicon Gynecare Prolift vaginal mesh, including chronic pain, mesh contraction, mesh erosion, pain during sexual intercourse, recurring prolapse of organs and urinary incontinence. As a result of the complications, Ms. Bellew asserts that she has undergone at least four operations to have the mesh removed.
Share This


