Zimmer Holdings Shareholder Suit Dismissed — Implant Defect Suits Continue
When the Plumbers and Pipefitters local union of West Virginia invested in Zimmer Holdings, shareholders were looking to reap some big financial rewards from the medical device maker. Instead they found themselves initiating a class action lawsuit that was dismissed Monday, May 21, by a federal appeals court judge. The U.S. Court of Appeals for the Seventh Circuit ruled Zimmer did not mislead investors about its defective metal hip replacement device, the Durom Cup, used to replace the socket in a hip.
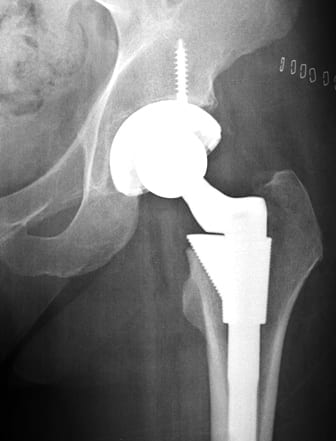
In January 2008, the company announced the Durom Cup would present a challenge to some surgeons as difficult to implant and that additional instructions and training might be necessary but Zimmer did not quantify the problem in a call with investors in January 2008, even as the company’s replacement components were recalled after finding they didn’t function properly. The judge ruled that can’t be treated as fraud.
Plaintiffs said that Zimmer also withheld quality-control problems at its plant in Dover, Ohio which it knew about as early as 2007. That information was not revealed during the January 2008 call with investors and three months later Zimmer closed some of its production lines for one month. By July 2008, the company cut revenue projections.
The fact that more problems with Zimmer came out over time did not imply that the company or its managers were lying to investors the judge ruled.
Multidistrict Litigation
The Durom Cup has been widely used as a hip replacement in the U.S. since 2006. However, when the hip replacement loosened, failed, and caused pain requiring repeat surgeries as well as metal poisoning, product liability cases were centralized by the US Judicial Panel on Multidistrict Litigation into MDL No. 2158 in the New Jersey District Court.
An estimated 12,000 people have been implanted with the Zimmer Durom Cup, despite the warning of inadequate instructions and early failures. Sales were suspended in July 2008 after reports that many patients suffering complications, had to undergo painful revision surgeries.
At least 75 cases are consolidated and pre-trial proceedings are underway. Expect the first bellwether trial to begin this year.
Zimmer has been settling lawsuits before they go to trial and started out in 2008 with a settlement fund of $47.5 million that had grown to $145 million by 2010.
The U.S. Food and Drug Administration will convene an expert panel June 27 and 28 to seek clinical opinions on the metal-on-metal (MOM) hip systems. Specifically the panel will be asked to focus on:
- Failure rates and causes;
- Metal (cobalt and chromium) ion testing;
- Imaging methods for failure detection;
- Local and systemic complications of implant failure;
- Patient risk factors; and
- Considerations for follow-up after surgery.
Share This


